Cannabis Market Transition: A 4/20 Reality Check for Investors
April 20 is the cannabis community’s day of symbolic celebration, and this year the date carries the weight of a definitive federal ultimatum.
Following US President Donald Trump’s December executive order to “expeditiously” move cannabis to Schedule III, the view from the executive suite is one of cautious pragmatism rather than unbridled triumph.
Rescheduling process introduces further uncertainty
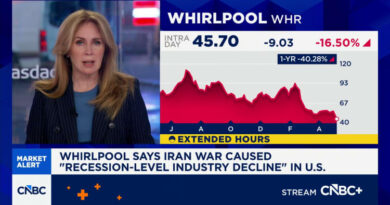
In the days following the signing of an executive order directing the Attorney General to complete the rulemaking process to reschedule cannabis from Schedule I to Schedule III of the Controlled Substances Act “in the most expeditious manner”, cannabis stocks saw some of their largest single-day gains in years.
The rally was driven by the realization that moving to Schedule III would effectively end the IRC Section 280E tax penalty, which currently prevents cannabis companies from deducting normal business expenses.
The president issued the executive order in December 2025 following the passing of a negotiated funding bill by the House and Senate Appropriations Committees, which removed a previous House provision designed to block the administration from using federal funding to reschedule cannabis.
However, a more sober reality has replaced that initial high, as the status of cannabis rescheduling remains pending amidst legislative attempts to block the move. A coalition of lawmakers, including House Speaker Mike Johnson, has also voiced formal opposition.
The current political climate followed a chaotic episode in 2025, where the DEA’s Chief Administrative Law Judge, John Mulrooney II, accused the agency of failing to provide a basic list of witnesses and evidence and abruptly cancelled the evidentiary hearings just days before they were set to begin.
Meanwhile, the Centers for Medicare & Medicaid Services recently launched a pilot program to cover certain hemp-derived CBD products.
The Substance Access Beneficiary Engagement Incentive allows healthcare providers in certain models, like the Enhancing Oncology Model, to offer eligible hemp-derived CBD products to patients. The program caps product value at US$500 per year and excludes inhalables, but represents the first time a federal agency has created a structured pathway for cannabinoid access within the Medicare framework.
While this marks a historic step toward federal medical integration, it arrives just as the broader industry faces a regulatory cliff.
Under a provision passed within the funding bill in November 2025, the federal government is moving toward a total THC limit of 0.4mg per container, a standard that would effectively recriminalize the majority of hemp-derived gummies and drinks currently on the market.
The provision established a one-year grace period before enforcement begins. The November 2026 deadline serves as the final cutoff for manufacturers to either remove these products from shelves or face…